I used to think a clinical data management course would be mostly about learning software. Like you join, learn a few tools, and you’re ready to work. But after digging into it a bit, it feels more layered than that.
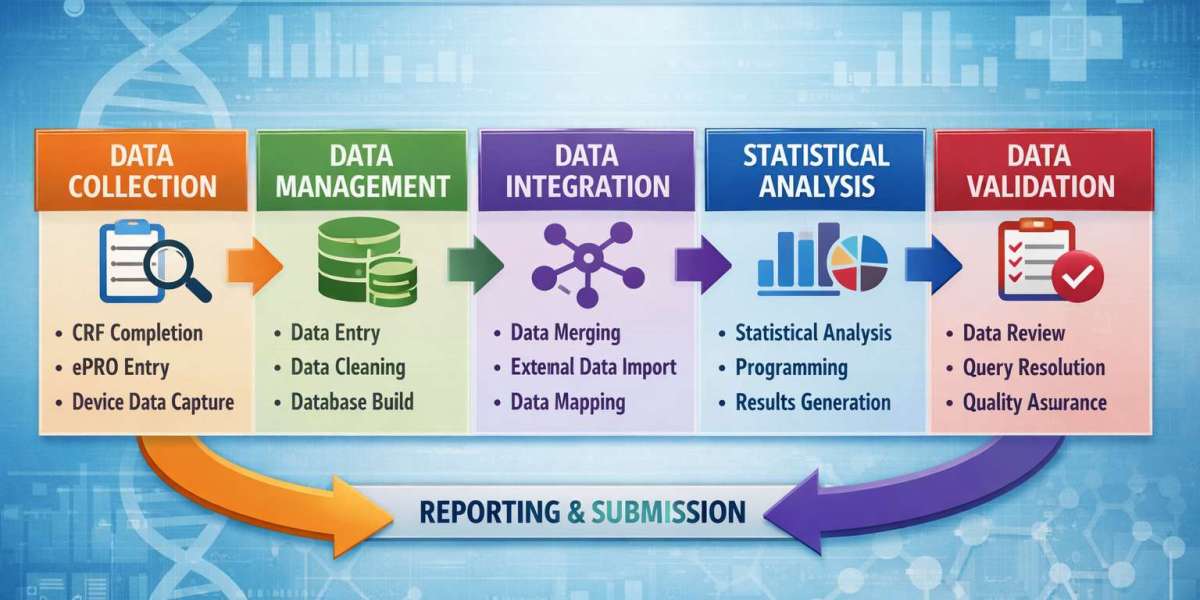
The structure of a clinical data management course isn’t always obvious from the outside. Most institutes list modules like “clinical research basics,” “data management,” “validation,” and “regulatory guidelines.” But what those modules really involve depends a lot on how the course is taught.
For example, the first part usually covers clinical research fundamentals. At first, I didn’t see why that was necessary. I thought, “I just want to work with data, not study the whole research process.”
But later, it made more sense.
If you don’t understand how a clinical trial is designed — phases, protocols, patient data collection — then the data itself doesn’t mean much. You’re just handling numbers without context.
A proper clinical data management course seems to start from that foundation. It builds up from understanding trials to understanding how data is captured and managed.
Then comes the tools part, which is what most people are curious about.
Tools like Medidata Rave or Oracle Clinical are often mentioned. But from what I’ve seen, not every course gives full hands-on access. Some just demonstrate workflows. Others allow practice, which is a big difference.
I spoke to someone who had completed a course recently. He said the real learning started when he began working on mock datasets. Before that, everything felt theoretical.
That stuck with me because it shows that tools alone aren’t enough. You need scenarios where things go wrong — missing data, inconsistent entries, validation errors — and you learn how to fix them.
That’s probably the closest you get to actual work during training.
Another part of the clinical data management course structure is understanding regulatory standards. This is where it gets slightly heavy.
Guidelines like Good Clinical Practice (GCP) aren’t just concepts. They define how data should be handled, documented, and verified. I tried reading about GCP through the World Health Organization (WHO) resources, and it made me realize how strict the industry is about compliance.
It’s not just about doing the job, it’s about doing it correctly and consistently.
Career scope is something that everyone asks about, but I think it’s often oversimplified.
Yes, there are roles like Clinical Data Coordinator, Data Manager, or even Clinical Research Associate if you move laterally. But getting into those roles depends on more than just completing a course.
Your understanding, your ability to handle tools, and how well you grasp real-world scenarios all matter.
I found a helpful explanation while going through this page (see details: https://www.hrremedyindia.com/clinical-data-management-courses-in-pune/), which breaks down how training is aligned with job roles. It didn’t guarantee anything, but it clarified expectations.
One thing I’ve noticed is that people sometimes underestimate the effort required after the course.
Just finishing a clinical data management course doesn’t automatically make you confident with data workflows. You need to revisit concepts, practice tools, and sometimes even learn additional things like basic Excel functions or data logic.
HR Remedy India often comes up as an example of a place learners often look at for practical, job-oriented exposure, especially when people discuss structured training. But like any institute, what you get depends on how much you engage with the course.
Another interesting observation — the pace of the course can affect how much you actually absorb.
Fast-paced courses might help you finish quickly, but you may not fully understand workflows. Slower courses give more time, but require patience.
I personally feel that a balanced approach works better. Enough time to practice, but not so slow that you lose momentum.
Also, the way instructors handle doubts matters more than I expected. Clinical data management involves small details, and if those aren’t clarified properly, confusion builds up.
So when evaluating a clinical data management course, I think it’s less about the module list and more about how those modules are delivered.
Are you just listening, or actually doing?
Are you memorizing terms, or understanding processes?
Because in the end, the role involves responsibility. You’re dealing with clinical trial data that can impact decisions in healthcare.
And that’s not something you want to approach casually.