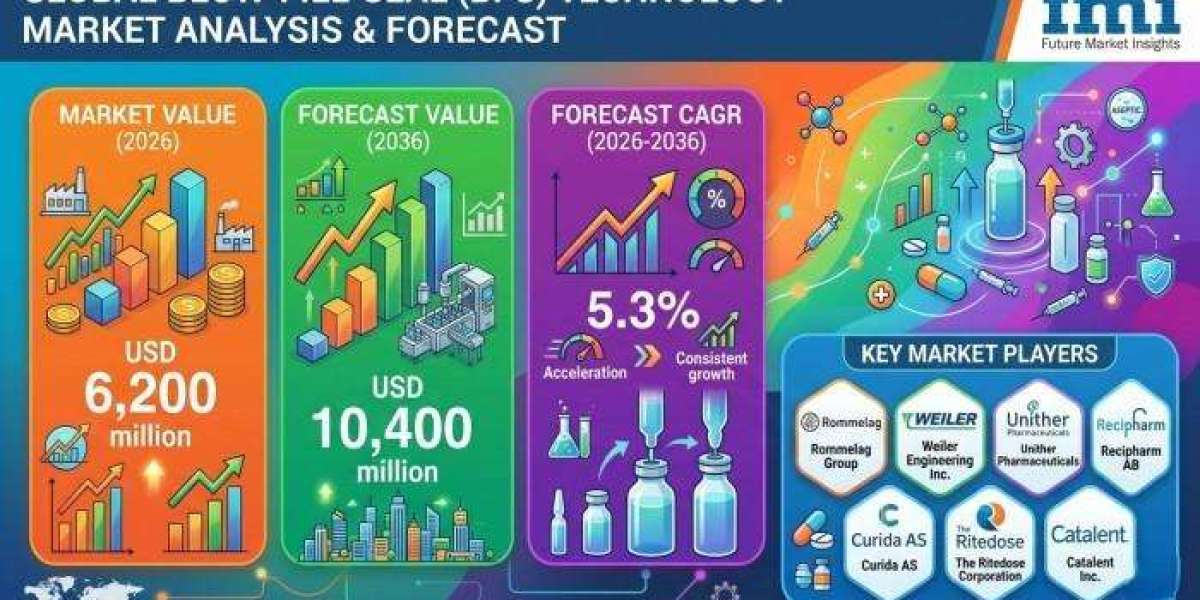
According to the latest market analysis by Future Market Insights, the blow-fill-seal (BFS) technology market is evolving into a critical infrastructure layer for sterile pharmaceutical packaging. Valued at USD 5,300 million in 2025 and projected to reach USD 6,200 million in 2026, the market is expected to expand at a CAGR of 5.3%, ultimately reaching USD 10,400 million by 2036.
This growth reflects a structural transition in pharmaceutical packaging—from multi-step, contamination-prone filling systems toward integrated, aseptic, and high-efficiency single-dose production platforms.
Quick Stats at a Glance
- Market size (2025): USD 5.3 billion
- Estimated value (2026): USD 6.2 billion
- Forecast (2036): USD 10.4 billion
- CAGR (2026–2036): 5.3%
- Incremental opportunity: USD 4.2 billion
- Leading application: Ophthalmic packaging (37% share)
- Top container type: Ampoules
- Dominant material: Polyethylene (54%+ share)
Get detailed market forecasts, competitive benchmarking, and pricing trends:
https://www.futuremarketinsights.com/reports/sample/rep-gb-4407
Market Size and Structural Shift
The blow-fill-seal technology market is entering a steady expansion phase, supported by rising demand for sterile, contamination-resistant packaging formats. However, beyond growth numbers, the real transformation lies in how pharmaceutical companies are redesigning packaging operations.
Traditional glass vial filling involves multiple stages—washing, sterilization, filling, and sealing—each introducing contamination risks and operational complexity. BFS technology eliminates these steps by forming, filling, and sealing containers within a closed sterile system.
This shift is enabling:
- Reduced contamination risks
- Higher production efficiency
- Simplified logistics
- Cost optimization in high-volume sterile manufacturing
Growth Drivers: Sterility, Efficiency, and Healthcare Demand
- Rising Demand for Preservative-Free Therapies
Global production of preservative-free ophthalmic drugs is increasing rapidly, driving adoption of BFS ampoule packaging that ensures sterile, single-dose delivery.
- Expansion of Respiratory Therapy Manufacturing
Localization of nebulizer solution production—especially in Asia—is accelerating demand for BFS-based unit-dose packaging systems.
- Hospital Shift Toward Ready-to-Use Formats
Healthcare systems are increasingly adopting pre-filled saline flush and irrigation solutions, reinforcing BFS deployment across hospital supply chains.
Market Constraints: Qualification Complexity and Material Sensitivity
Despite strong adoption trends, BFS technology faces certain operational barriers:
- Extractables and leachables validation: Stringent regulatory requirements for polymer-based containers
- Oxygen permeability concerns: Limits use in biologics and sensitive drug formulations
- High installation and validation costs: Particularly for advanced sterile manufacturing setups
As a result, BFS adoption remains strongest in liquid pharmaceuticals where stability and sterility requirements align with polymer packaging capabilities.
Opportunity Landscape: Where Growth Is Emerging
Several high-impact opportunities are shaping the future of BFS deployment:
- Single-dose packaging expansion: Increasing shift away from multi-dose formats
- Polymer container adoption: Transition from glass to lightweight, break-resistant materials
- Contract manufacturing growth: CDMOs expanding BFS capabilities for flexible production
- Hospital supply chain modernization: Demand for ready-to-use injectable formats
Segment Insights: Where the Market Is Concentrated
By Product Type:
Ampoules dominate the market due to their compatibility with ophthalmic and respiratory therapies requiring precise, single-dose delivery.
By Material Type:
Polyethylene, particularly LDPE, leads with over 54% share, offering:
- High compatibility with sterile formulations
- Lightweight and break-resistant properties
- Efficient extrusion processing
By Container Size:
Containers below 0.2 L hold the largest share, widely used in:
- Eye-drop formulations
- Nebulizer solutions
- Saline flush applications
By End Use:
Ophthalmic applications lead with a 37% share, driven by global demand for preservative-free sterile drug delivery.
Regional Dynamics: Growth Led by Asia Pacific
Asia Pacific – Primary Growth Engine
- India (8.2% CAGR): Expansion of ophthalmic export manufacturing and respiratory therapy localization
- China (7.4% CAGR): Strong sterile generic manufacturing capacity and regulatory alignment
Latin America – Emerging Opportunity
- Brazil (6.3% CAGR): Growth driven by hospital procurement modernization and sterile packaging demand
North America – Mature but Stable
- United States (4.9% CAGR): Growth supported by hospital adoption of ready-to-use injectable formats
Europe – Innovation and Compliance Hub
- Germany (4.4% CAGR): Focus on sterile manufacturing upgrades and ophthalmic packaging infrastructure
Competitive Landscape: High Entry Barriers, Strong Specialization
The BFS market is moderately consolidated, dominated by specialized aseptic equipment providers and pharmaceutical CDMOs.
Key competitive differentiators include:
- Multi-cavity tooling capability
- Sterile enclosure validation expertise
- Compatibility with diverse pharmaceutical applications
- Regulatory compliance readiness
High switching costs and stringent validation requirements create strong entry barriers, reinforcing long-term supplier relationships.
Strategic Implications for Industry Stakeholders
- Manufacturers: Prioritize BFS for ophthalmic and respiratory packaging to improve sterility and efficiency
- Healthcare providers: Align procurement strategies with ready-to-use sterile packaging formats
- Investors: Focus on CDMOs and equipment suppliers with BFS specialization
- Procurement leaders: Evaluate suppliers based on compliance, reliability, and scalability
Future Outlook: From Adoption to Standardization
Over the next decade, BFS technology is expected to become a standard for liquid sterile pharmaceutical packaging.
Key trends to watch:
- Expansion of polymer-based sterile packaging
- Increased CDMO outsourcing for BFS production
- Growth in single-dose drug delivery systems
- Integration of automated, high-throughput BFS platforms
As pharmaceutical manufacturing continues to prioritize sterility, efficiency, and scalability, BFS technology will play a central role in shaping next-generation packaging infrastructure.
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.